Brining chickens before you smoke them is supposed to give them better texture and to leave them more moist after cooking. Several times I have tried bring chickens before smoking and on each occasion I have either not noticed any difference in the end result or they have come out too salty. None of these brines though have been true comparative tests and so I thought it was time to directly compare a number of common home brining methods. As well as any texture and moisture improvement, one of the things I was hoping to establish was whether some of the published methods of scientifically calculating the brine update process were accurate (or even possible). Also whether brining chickens was actually more an art than a science.
After looking at the various methods other people have been using successfully (on here and on other forums and chefs sites) I decided to directly compare the following three brining methods.
Having reviewed the optimal brine strengths mentioned in different sources, and as we are quite sensitive to salt in foods, I decided to try for an end result of 1% salt in the chicken. This being balanced by 0.5% sugar.
The methods I decided to use for each type of brine were based upon those described on the Stella Culinary site video lectures about the science behind brining.
https://stellaculinary.com/cooking-...cience-behind-brining-four-part-video-lecture
What I describe below is the theory of what is supposed to happen, and where I ran into difficulties I have highlighted these as observations
All of the chickens were purchased on the same day from the same supplier and were of similar weights (approx 1.6 Kg - 3.5 pounds). They had not been pre-processed or previously brined.
The calculations used for calculating the amount of brine and brine concentrations were based upon the meat weights of each chicken as the bone does not readily absorb the brine. The calculation used make the assumption that 40% of the weight of the bird was bone - therefore 60% is meat.
The different brines. Below I am describing the theory as described behind each method.
Dry Brine
This works in the same way as dry curing bacon. The amount of brine/cure is calculated from the weight of the meat which it is applied to the surface and left to diffuse into the meat.
For this the salt was weighed out to give 1% the weight of the chicken meat and the sugar for 0.5%. These were mixed and rubbed over the surfaces of the chicken - both inside and out.
Gradient Brine
This method works by creating a 5% brine (which is stronger than final desired salt concentration) and allowing it to diffuse into the meat. The chicken is removed from the brine when sufficient salt has been absorbed from the brine so that when it has been allowed to rest the salt concentration will equalise at the final desired concentration. This method is supposed to require less time for the chicken in the brine however it requires standing time once it has been brined. The amount of brine absorbed by the meat is calculated by the loss of the salt from the surrounding brine solution. Once the concentration in the brine has reduced by 1% then ~10 g of salt will have entered the chicken and the final salt in the meat should be 1%. The weight of brine used was equal to the calculated weight of the meat. The brining time I intended to use was that recommended by Stella Culinary for whole chickens - 24-48 hours in the brine with an 8-24 hour rest time.
Equillibrium Brine
This method works by creating a brine that is 2x stronger than final desired salt concentration and using the same weight of brine as there is meat. The chicken is removed from the brine when the surrounding brine concentration has reduced by 50% indicating that 50% of the salt has diffused into the meat. The weight of brine used was equal to the calculated weight of the meat.
Control
This was bought, stored and cooked under the same conditions to see if simply storing time would affect the chicken
Chicken weights and brine concentrations
How to measure the brine concentration
As we are trying to measure the salt uptake be measuring the salt depletion in the brine it was important to be able to accurately measure the brine salt concentrations. I have two different ways of measuring this - a Sodium ion salt meter and a salt refractometer
Firstly I wanted to check their accuracy on the brines and also whether the sugar would effect the salt measurements. I therefore measured the brines as they were being made, before and after the sugar was added.
The first thing I noticed was that with just the salt the Salt Meter was measuring within 2% and within 6% of the calculated concentrations. The refractometer was measuring within 5% and 9%.
Whilst, adding the sugar to the brine only had a minimal effect on the Salt meter, the effect on the refractometer was was significantly greater - nearly 50% for the 5%/2.5% brine and nearly 90% for the 2%/1% brine.
Observation - Unless the brine only contains salt (i.e. contains no sugar) you cannot use a refractometer to accurately measure the actual brine salt concentration. It may be possible if you are only trying to monitor relative concentrations - though I cannot tell from this whether the inaccuracy is in linear proportion to the sugar concentration, and so this would need more testing.
The brining process
The brines needed to completely cover the chickens and so as volumes of brine were quite small this was done by brining them in plastic bags in the fridge. The bags containing liquid brine had all of the air removed and were sealed using a zip lock. Approximately every 6 hours the bags were opened and the bags rotated vertically to ensure that the brine in the chicken cavity was thoroughly mixed with the brine surrounding the chicken. The air was then removed from the bag, the bags sealed and then placed back in the fridge.
Approximately every 9-12 hours a small sample of the brine was taken and the salt concentration measured
Although in the graph I have included the results from both the Salt Meter and the Refractometer, we should ignore the refractometer results due to the affect of the sugar. The only lines to look at in the graph are the red line (meter reading for 5% salt) and the yellow line (meter reading for 2% salt).
Observations
Both of the Gradient brining and equilibrium brining methods relied on measuring the reduction in salt in the brine to determine the uptake of salt into the chicken.
This would have required the 5% brine to have reduced in absolute concentrations by 1 % - which it did after 27 hours. This actually supports the recommended 24-48 hours in the brine.
The 2% brine also only needed to drop in absolute concentration by 1%, however after over 70 hours it was in the brine it had only dropped by 0.44%
When brining chicken it would appear that one of two things is happening, Either the salt does is not diffused throughout the chicken in the way that was described or there is something that is being released from the chicken that is interfering with both the Salt Meter and refractometer readings. It is likely that the difficulty with the equilibrium brine is that it is generally recognised that meat comprises of 75% water and this would need to be taken into account when calculating the required brine strength.
Due to the inability to adequately measure the uptake of the brine into the meat in the 2% brine and also the duration over which the brining occurred it was difficult to tell whether we were actually reaching the "equilibrium" brining - as defined by Stella Culinary.
Chicken and brine sample after time in brine
The cooking and eating
The chickens were smoked at 300F (150 C) in my FEC using hickory pellets until the IT of the birds reached 160 F (72 C)
One of the breasts was taken from each for the blind tasting panel to judge.
5% and 2% chickens
Dry Brine and control
The tasting panel were then asked to rank the samples on the following 3 criteria
The ranking was from 1-4, with 1 being the beat.
Conclusions
The different brining techniques had little overall effect on the overall eatability of the meat in this trial. The control (unbrined) chicken was ranked equally as enjoyable as the other chickens.
The brining did make a marginal difference to the moistness of the chicken, with the 5% and 2% brines
The 5% brine resulted in a 6% increase in weight whereas the dry and 2% brines resulted in a 2% increase. This additional 4% is likely to additional water taken up into the chicken.
* The additional saltiness in the 5% brined chicken was probably due to the chicken being left in the brine for longer than the 27 hours it actually took for it to reach a 1% salt uptake.
From the observations in this trial, I have to question the scientific explanations surrounding the calculations for both the Gradient and Equilibrium brining. The described reduction in salt from the brine as it diffused into the chicken just did not happen in the equillibrium brine. This is not really surprising as the diffusion assumptions would have to assume that the brine salt will reach an equal concentration equilibrium between brine fluid and the meat. This may be true if the meat was approaching 100% water and the salt could diffuse equally throughout it - however this is not the case.
At least using this methodology the scientific approach to determining when a chicken has been suitably brined was not supported for the Equilibrium brine. It is likely that by better understanding the way that the brine diffuses within the meat we would be able to develop a more accurately predict model for the brining process - but until then I have to conclude that getting the perfect brine in a chicken is more of an art than a science.There is nothing wrong with this and it gives more confidence to try different recipes that have evolved different brining techniques.
There is another method of brining here that I did not cover in the trial, and that is injection brining. This method would likely move the balance back in favour of science and is why it is used in many commercially brined meats.
*** Update - A special thanks to wbf610 who pointed out an error in my 5% brine calculation. The absolute concentration of salt in the brine only needed to drop by 1% for the chicken to reach 1% salt and not the 2% as originally stated. This means that the chicken in the 5% brine did reach the calculated brine reduction after about 27-28 hours. This means that it would actually be possible to use the 5% brine to calculate when the chicken had taken up sufficient salt. This still does not appear to be the case when it comes to the 2% equilibrium brine though. The text of the post has therefore been amended slightly to reflect this.
After looking at the various methods other people have been using successfully (on here and on other forums and chefs sites) I decided to directly compare the following three brining methods.
- Dry brine
- Gradient brine
- Equilibrium brine
Having reviewed the optimal brine strengths mentioned in different sources, and as we are quite sensitive to salt in foods, I decided to try for an end result of 1% salt in the chicken. This being balanced by 0.5% sugar.
The methods I decided to use for each type of brine were based upon those described on the Stella Culinary site video lectures about the science behind brining.
https://stellaculinary.com/cooking-...cience-behind-brining-four-part-video-lecture
What I describe below is the theory of what is supposed to happen, and where I ran into difficulties I have highlighted these as observations
All of the chickens were purchased on the same day from the same supplier and were of similar weights (approx 1.6 Kg - 3.5 pounds). They had not been pre-processed or previously brined.
The calculations used for calculating the amount of brine and brine concentrations were based upon the meat weights of each chicken as the bone does not readily absorb the brine. The calculation used make the assumption that 40% of the weight of the bird was bone - therefore 60% is meat.
The different brines. Below I am describing the theory as described behind each method.
Dry Brine
This works in the same way as dry curing bacon. The amount of brine/cure is calculated from the weight of the meat which it is applied to the surface and left to diffuse into the meat.
For this the salt was weighed out to give 1% the weight of the chicken meat and the sugar for 0.5%. These were mixed and rubbed over the surfaces of the chicken - both inside and out.
Gradient Brine
This method works by creating a 5% brine (which is stronger than final desired salt concentration) and allowing it to diffuse into the meat. The chicken is removed from the brine when sufficient salt has been absorbed from the brine so that when it has been allowed to rest the salt concentration will equalise at the final desired concentration. This method is supposed to require less time for the chicken in the brine however it requires standing time once it has been brined. The amount of brine absorbed by the meat is calculated by the loss of the salt from the surrounding brine solution. Once the concentration in the brine has reduced by 1% then ~10 g of salt will have entered the chicken and the final salt in the meat should be 1%. The weight of brine used was equal to the calculated weight of the meat. The brining time I intended to use was that recommended by Stella Culinary for whole chickens - 24-48 hours in the brine with an 8-24 hour rest time.
Equillibrium Brine
This method works by creating a brine that is 2x stronger than final desired salt concentration and using the same weight of brine as there is meat. The chicken is removed from the brine when the surrounding brine concentration has reduced by 50% indicating that 50% of the salt has diffused into the meat. The weight of brine used was equal to the calculated weight of the meat.
Control
This was bought, stored and cooked under the same conditions to see if simply storing time would affect the chicken
Chicken weights and brine concentrations
| Chicken | Brine type | Initial weight (g) | "Boneless" weight (g) | Water (g) | Salt (G) | Sugar (G) | estimated Time in brine | Rinse after brining | Weight after brining | % weight increase | Stand | Smell after brining |
| 1 | Dry brine 1% | 1,687 | 1,012 | 0 | 10 | 5 | 36 hours | NO | 1,724 | 2 | 12 hours | None |
| 2 | Gradient 5% | 1,642 | 985 | 911 | 49 | 25 | 36 hours | YES | 1,740 | 6 | 24 hours | None |
| 3 | Equilibrium 2% | 1,649 | 989 | 960 | 20 | 10 | ? | NO | 1,680 | 2 | 12 hours | None |
| 4 | Control | 1,578 | 947 | 0 | 0 | n/a | NO | 1,577 | 0 | Faint pea smell |
As we are trying to measure the salt uptake be measuring the salt depletion in the brine it was important to be able to accurately measure the brine salt concentrations. I have two different ways of measuring this - a Sodium ion salt meter and a salt refractometer
Firstly I wanted to check their accuracy on the brines and also whether the sugar would effect the salt measurements. I therefore measured the brines as they were being made, before and after the sugar was added.
| Water | Salt | Calculated Salt % | Measured % Salt meter | Measured % Refractometer | Sugar | Measured % Salt meter | Measured % Refractometer | ||
| 5% brine | 911 | 49 | 5.10 | 5.20 | 5.20 | 25 | 5.20 | 7.40 | |
| 2% brine | 960 | 20 | 2.04 | 2.16 | 2.11 | 10 | 2.20 | 3.80 |
Whilst, adding the sugar to the brine only had a minimal effect on the Salt meter, the effect on the refractometer was was significantly greater - nearly 50% for the 5%/2.5% brine and nearly 90% for the 2%/1% brine.
Observation - Unless the brine only contains salt (i.e. contains no sugar) you cannot use a refractometer to accurately measure the actual brine salt concentration. It may be possible if you are only trying to monitor relative concentrations - though I cannot tell from this whether the inaccuracy is in linear proportion to the sugar concentration, and so this would need more testing.
The brining process
The brines needed to completely cover the chickens and so as volumes of brine were quite small this was done by brining them in plastic bags in the fridge. The bags containing liquid brine had all of the air removed and were sealed using a zip lock. Approximately every 6 hours the bags were opened and the bags rotated vertically to ensure that the brine in the chicken cavity was thoroughly mixed with the brine surrounding the chicken. The air was then removed from the bag, the bags sealed and then placed back in the fridge.
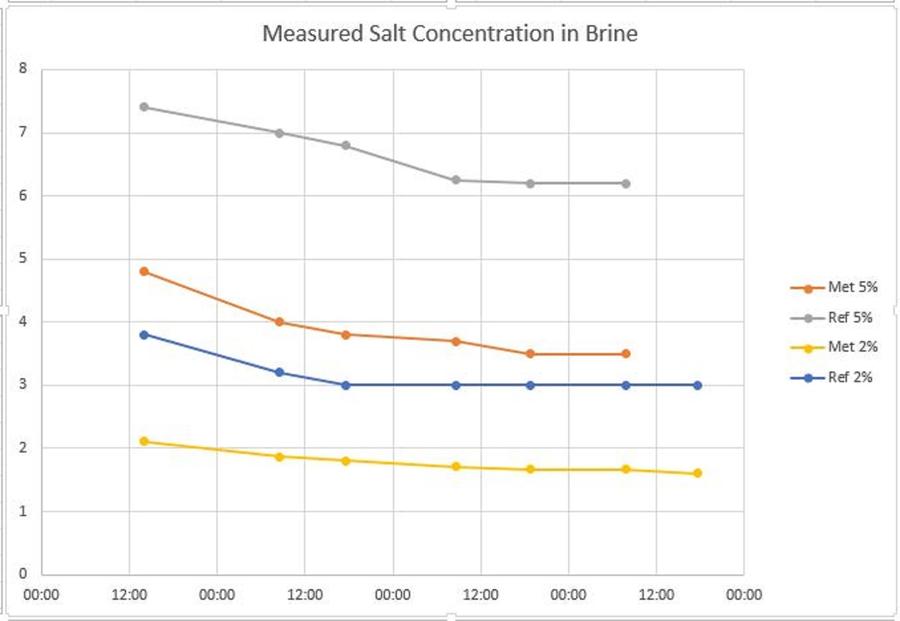
Approximately every 9-12 hours a small sample of the brine was taken and the salt concentration measured
| Time | Time in Brine hh:mm | Salt meter 5% | Refractometer 5% | Salt meter 2% | Refractometer 2% |
| 23/10/2016 14:00 | 4.8 | 7.4 | 2.11 | 3.8 | |
| 24/10/2016 08:30 | 18:30 | 4 | 7 | 1.87 | 3.2 |
| 24/10/2016 17:30 | 27:30 | 3.8 | 6.8 | 1.8 | 3 |
| 25/10/2016 08:30 | 42:30 | 3.7 | 6.25 | 1.71 | 3 |
| 25/10/2016 18:45 | 52:45 | 3.5 | 6.2 | 1.67 | 3 |
| 26/10/2016 07:45 | 64:45 | 3.5 | 6.2 | 1.67 | 3 |
| 26/10/2016 17:30 | 73:30 | 1.6 | 3 |
Although in the graph I have included the results from both the Salt Meter and the Refractometer, we should ignore the refractometer results due to the affect of the sugar. The only lines to look at in the graph are the red line (meter reading for 5% salt) and the yellow line (meter reading for 2% salt).
Observations
Both of the Gradient brining and equilibrium brining methods relied on measuring the reduction in salt in the brine to determine the uptake of salt into the chicken.
This would have required the 5% brine to have reduced in absolute concentrations by 1 % - which it did after 27 hours. This actually supports the recommended 24-48 hours in the brine.
The 2% brine also only needed to drop in absolute concentration by 1%, however after over 70 hours it was in the brine it had only dropped by 0.44%
When brining chicken it would appear that one of two things is happening, Either the salt does is not diffused throughout the chicken in the way that was described or there is something that is being released from the chicken that is interfering with both the Salt Meter and refractometer readings. It is likely that the difficulty with the equilibrium brine is that it is generally recognised that meat comprises of 75% water and this would need to be taken into account when calculating the required brine strength.
Due to the inability to adequately measure the uptake of the brine into the meat in the 2% brine and also the duration over which the brining occurred it was difficult to tell whether we were actually reaching the "equilibrium" brining - as defined by Stella Culinary.
Chicken and brine sample after time in brine
The cooking and eating
The chickens were smoked at 300F (150 C) in my FEC using hickory pellets until the IT of the birds reached 160 F (72 C)
One of the breasts was taken from each for the blind tasting panel to judge.
5% and 2% chickens
Dry Brine and control
The tasting panel were then asked to rank the samples on the following 3 criteria
- Saltiness
- Moistness
- Overall eating experience
The ranking was from 1-4, with 1 being the beat.
| 5% Brine | 2% Brine | Dry Brine | Control | Comments | |
| Saltiness | 4* | 1 | 3 | 1 | The 5% brine was mildly over salted but was not unpleasant |
| Moistness | 1 | 1 | 3 | 4 | All were nice and moist however a slight preference was shown towards the brined chickens |
| Overall eating | 4 | 1 | 1 | 1 | All were enjoyed however the higher salt in the 5% brine made it the least favourite. |
The different brining techniques had little overall effect on the overall eatability of the meat in this trial. The control (unbrined) chicken was ranked equally as enjoyable as the other chickens.
The brining did make a marginal difference to the moistness of the chicken, with the 5% and 2% brines
The 5% brine resulted in a 6% increase in weight whereas the dry and 2% brines resulted in a 2% increase. This additional 4% is likely to additional water taken up into the chicken.
* The additional saltiness in the 5% brined chicken was probably due to the chicken being left in the brine for longer than the 27 hours it actually took for it to reach a 1% salt uptake.
From the observations in this trial, I have to question the scientific explanations surrounding the calculations for both the Gradient and Equilibrium brining. The described reduction in salt from the brine as it diffused into the chicken just did not happen in the equillibrium brine. This is not really surprising as the diffusion assumptions would have to assume that the brine salt will reach an equal concentration equilibrium between brine fluid and the meat. This may be true if the meat was approaching 100% water and the salt could diffuse equally throughout it - however this is not the case.
At least using this methodology the scientific approach to determining when a chicken has been suitably brined was not supported for the Equilibrium brine. It is likely that by better understanding the way that the brine diffuses within the meat we would be able to develop a more accurately predict model for the brining process - but until then I have to conclude that getting the perfect brine in a chicken is more of an art than a science.There is nothing wrong with this and it gives more confidence to try different recipes that have evolved different brining techniques.
There is another method of brining here that I did not cover in the trial, and that is injection brining. This method would likely move the balance back in favour of science and is why it is used in many commercially brined meats.
*** Update - A special thanks to wbf610 who pointed out an error in my 5% brine calculation. The absolute concentration of salt in the brine only needed to drop by 1% for the chicken to reach 1% salt and not the 2% as originally stated. This means that the chicken in the 5% brine did reach the calculated brine reduction after about 27-28 hours. This means that it would actually be possible to use the 5% brine to calculate when the chicken had taken up sufficient salt. This still does not appear to be the case when it comes to the 2% equilibrium brine though. The text of the post has therefore been amended slightly to reflect this.
Last edited: